Many chemical compounds have versatile uses. Sometimes, drugs which were developed for a certain purpose are surprisingly useful in other areas. Tamoxifen (tuh-MOCKS-if-en) is one such drug. Perhaps you have heard of it; you someone you know may have been treated with it. This compound is an effective treatment against certain common types of breast cancer.
Tamoxifen, or more specifically the Z isomer of tamoxifen, is an estrogen receptor antagonist.What that means is that tamoxifen will stick to estrogen receptors in the body and exclude estrogen from binding. Thanks to this receptor specificity, tamoxifen accumulates in estrogen receptor-positive breast cancer cells 200-fold more than in healthy cells. Tamoxifen’s natural affinity for estrogen receptor positive breast cancer cells means you can achieve lethal doses in cancer cells while not killing the normal, healthy cells. Chemotherapeutic drugs are often equally as toxic to normal cells as they are to cancer cells, so to minimize toxic side effects, concentrating the drug in the target area is crucial. For this reason, most cancer research is focused on targeting and delivery.
In addition to traditional cancer treatment, tamoxifen has found a particular niche as an adjuvant, or preventative, therapy. It is fairly common for women who have undergone successful breast cancer therapy to relapse. Adjuvant therapy involves indefinite treatment with a post-operative chemopreventative agent, such as tamoxifen. Women who are regularly administered tamoxifen after breast cancer treatments have a much lower rate of relapse. Tamoxifen is generally used in an adjuvant manner in post-menopausal women but not in pre-menopausal women, because of side effects precipitated by tamoxifen’s effect on estrogen receptors. Tamoxifen, then, is an extremely useful compound in the sphere of breast cancer treatment. But what if this common drug was capable of much, much more?
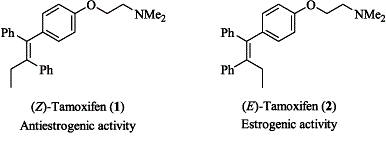
Tamoxifen is an interesting compound. It is hydrophobic (water-hating, like oil) except for a small tail which contains a nitrogen. This nitrogen is called a terminal tertiary amine. Terminal because it is at the end of the chain, tertiary because it is attached to three other things that are not hydrogen, and amine because it is nitrogen single-bonded only to carbon, which you can see in the above picture. When I was working on my thesis at Georgia Tech, I altered this end of the molecule, attaching different chemical groups in the hopes of increasing tamoxifen’s antibacterial activity. Yes, in addition to being used in chemotherapy and adjuvant treatments, tamoxifen also shows antibacterial activity.
Though the exact mechanism has not been elucidated, tamoxifen likely disrupts bacterial cell membranes, causing them to become leaky. When this membrane is compromised, cellular material can seep into the extracellular environment, leading to cell death. One observed effect is ion efflux, meaning that the ions within the cell like potassium and sodium, which are crucial to cell function, escape the bacteria into the surroundings. Without a proper balance of ions, the bacterium dies. The evidence for this comes from several images of bacterial cells before and after treatment with tamoxifen, where damage to the cell membrane can be seen after treatment, as well as quantified potassium ion efflux after treatment with tamoxifen. In short, tamoxifen poked holes in bacterial cell membranes, causing the cells to leak potassium, leading to cell death. All of this suggests that tamoxifen, an anticancer drug, may also be useful as an antibacterial drug. Other research shows that tamoxifen may be useful as an antifungal agent, could be used to treat leishmanial parasites, and is effective against tuberculosis which is resistant to other treatments.
Keep in mind that most of this research is in its infancy and it may be years before tamoxifen could be approved for anything besides cancer treatment. The point is to show that chemical compounds can often multitask in very interesting ways. Tamoxifen itself had an interesting history, initially being developed as birth control. Although it later found its calling as an anticancer agent and adjuvant therapy, tamoxifen was unsuccessful at the purpose for which it was created. In tests, tamoxifen actually induced ovulation. Some forward-thinking scientists realized the potential of the drug, however, and pushed it into cancer treatment trials, where it happily reigns today.


